Historical Evolution and Legislative Philosophy
The contemporary Indian patent regime emerged from a shift away from colonial-era laws toward a system aligned with the developmental needs of an independent nation. Before the Patents Act, 1970, intellectual property was governed by the Indian Patents and Designs Act, 1911, which largely favoured foreign corporations. These entities held nearly 80–90% of patents in India, yet only about 10% were worked locally, often blocking domestic competition and keeping prices especially of medicines high.
India’s accession to the World Trade Organization and compliance with the Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS) further reshaped its patent framework. Through amendments in 1999, 2002, and particularly 2005, India reintroduced product patents across all fields of technology while retaining safeguards to protect public health.
| Era / Event | Legislative Focus | Primary Outcome |
|---|---|---|
| Colonial Period (1911 Act) | Foreign corporate interests and market control | High drug prices; 90% foreign patent ownership |
| Ayyangar Committee (1950s) | Socioeconomic welfare and industrial growth | Foundation for the 1970 Act |
| 1970 Act (Original) | Process patents only for food and drugs | Rise of the Indian generic pharmaceutical industry |
| TRIPS Agreement (1995) | Global standardization of IP rights | Commitment to reintroduce product patents |
| 2005 Amendment | Full product patent regime and Section 3(d) safeguards | Alignment with international standards; anti-evergreening |
Statutory Definitions and the Threshold of Patentability
The effectiveness of the Patents Act, 1970, relies on its precise definition of an “invention” and the rigorous criteria used to evaluate patent applications. Under Section 2(1)(j), an invention is defined as a new product or process involving an inventive step and capable of industrial application. This definition serves as the primary filter for determining which technological advancements merit the 20 year period of market exclusivity.
The Triple Test of Patentability
For an innovation to be granted patent protection in India, it must satisfy three fundamental requirements: novelty, inventive step, and industrial applicability.
- Novelty: The subject matter must not have been anticipated by publication in any document or used anywhere in the world before the priority date. This ensures that patents are not granted for information already in the public domain or part of the “state of the art”.
- Inventive Step: According to Section 2(1)(ja), this represents a feature of an invention that involves technical advancement compared to existing knowledge or has economic significance, making the invention non-obvious to a person skilled in the art. The inclusion of “economic significance” is a distinctively Indian provision, allowing for patents where the primary innovation lies in cost reduction or resource efficiency rather than a massive technical leap.
- Industrial Applicability: The invention must be capable of being produced or used in an industry. Purely theoretical or abstract concepts that cannot be physically manifested or utilized in a commercial process are excluded.
The Act also clarifies the role of the “true and first inventor,” emphasizing that the right to apply for a patent belongs to the individual who actually conceived the invention, or their assignee. In instances where an application is based on an assignment, Rule 10 mandates that the applicant must provide documented proof of the transfer of rights.
Non-Patentable Subject Matter: The Section 3 Gatekeepers
Perhaps the most critical sections for innovators and businesses to understand are Sections 3 and 4, which enumerate subject matter that is expressly excluded from patentability. These provisions act as “gatekeepers,” ensuring that the patent system does not grant monopolies over basic scientific principles, traditional knowledge, or inventions that could harm public order or health.
Detailed Analysis of Section 3 Exclusions
The Act lists several categories of “non-inventions”:
- Section 3(a): Frivolous inventions or those that claim anything obviously contrary to well-established natural laws, such as a machine claiming to provide perpetual motion without energy input.
- Section 3(b): Inventions whose primary use or commercial exploitation could be contrary to public order or morality, or which cause serious prejudice to human, animal, or plant life, or the environment. This prevents the patenting of harmful devices or immoral technologies.
- Section 3(c): The mere discovery of a scientific principle, the formulation of an abstract theory, or the discovery of any living thing or non-living substance occurring in nature. For example, discovering a new planet or a pure natural substance does not constitute an invention.
- Section 3(d): The mere discovery of a new form of a known substance which does not result in the enhancement of the known efficacy of that substance. This provision is central to preventing “evergreening” in the pharmaceutical sector.
- Section 3(e): A substance obtained by a mere admixture resulting only in the aggregation of the properties of the components, such as a simple mixture of sugar and water.
- Section 3(f): The mere arrangement or re-arrangement or duplication of known devices, each functioning independently of one another in a known way.
- Section 3(h): Methods of agriculture or horticulture, such as a specific technique for growing wheat.
- Section 3(i): Any process for the medicinal, surgical, curative, prophylactic, diagnostic, therapeutic, or other treatment of human beings or animals to render them free of disease or to increase their economic value. This ensures that doctors and hospitals can treat patients without fearing patent infringement claims.
- Section 3(j): Plants and animals in whole or any part thereof, other than microorganisms, but including seeds, varieties, and species, and essentially biological processes for production or propagation of plants and animals.
- Section 3(k): A mathematical or business method or a computer programme per se or algorithms.
- Section 3(l): Literary, dramatic, musical, or artistic works or any other aesthetic creations, including cinematographic works and television productions, which are instead protected under Copyright Law.
- Section 3(m): A mere scheme or rule or method of performing a mental act or method of playing a game.
- Section 3(n): A presentation of information.
- Section 3(o): Topography of integrated circuits, which is protected under the Semiconductor Integrated Circuit Layout Design Act.
- Section 3(p): Inventions which, in effect, are traditional knowledge or which are an aggregation or duplication of known properties of traditionally known components.
| Section | Target of Exclusion | Policy Rationale |
|---|---|---|
| 3(d) | Incremental pharmaceutical changes | Prevents evergreening; ensures affordable medicine |
| 3(k) | Software and business models | Prevents monopoly on abstract logic and commerce |
| 3(p) | Traditional knowledge and bio piracy | Protects indigenous community rights |
| 3(i) | Medical treatment methods | Safeguards public health and doctor freedom |
The Battle Against Evergreening: Section 3(d) and the Novartis Precedent
Section 3(d) represents one of the most significant and controversial aspects of Indian patent law on the global stage. It stipulates that a new form of a known substance such as a salt, ester, ether, polymorph, or isomer is considered the same substance unless it differs significantly in properties regarding efficacy. This prevents pharmaceutical companies from filing a series of patents on minor variations of a drug to extend their monopoly beyond the initial 20-year term.
The definitive interpretation of this section came in the landmark case of Novartis AG v. Union of India (2013). Novartis challenged the rejection of its patent application for the beta-crystalline form of “Imatinib Mesylate,” an anti-cancer drug marketed as Gleevec. Novartis argued that the new crystalline form was more stable and had higher bioavailability than the known alpha form, thus qualifying as an invention. However, the Supreme Court of India ruled that “efficacy” in the context of pharmaceutical patents specifically means “therapeutic efficacy”. The Court held that improved physical properties like stability or solubility do not constitute enhanced therapeutic efficacy unless they result in a superior curative effect on the human body.
The implications of the Novartis decision are profound for both the industry and public health. It established a “floor” for pharmaceutical innovation, requiring applicants to demonstrate real and measurable improvements in treatment outcomes to secure secondary patents. This has allowed generic manufacturers to continue producing affordable versions of essential medicines, maintaining India’s role as a vital supplier of low-cost drugs to developing nations.
Digital Innovation and Section 3(k): Software and AI
The patentability of Computer-Related Inventions (CRIs) is governed by Section 3(k), which excludes “computer programmes per se“. The term “per se” was introduced through the 2002 amendment to distinguish between standalone software code and software that is part of a larger technical invention. The legislative intent was not to reject software patents entirely but to ensure that patents are only granted for software that achieves a “technical effect”.
The Indian Patent Office (IPO) released updated CRI Guidelines in 2025 to provide clarity in the rapidly evolving fields of Artificial Intelligence (AI), block chain, and quantum computing. These guidelines emphasize that examiners must look for a “technical contribution” that goes beyond abstract algorithms.
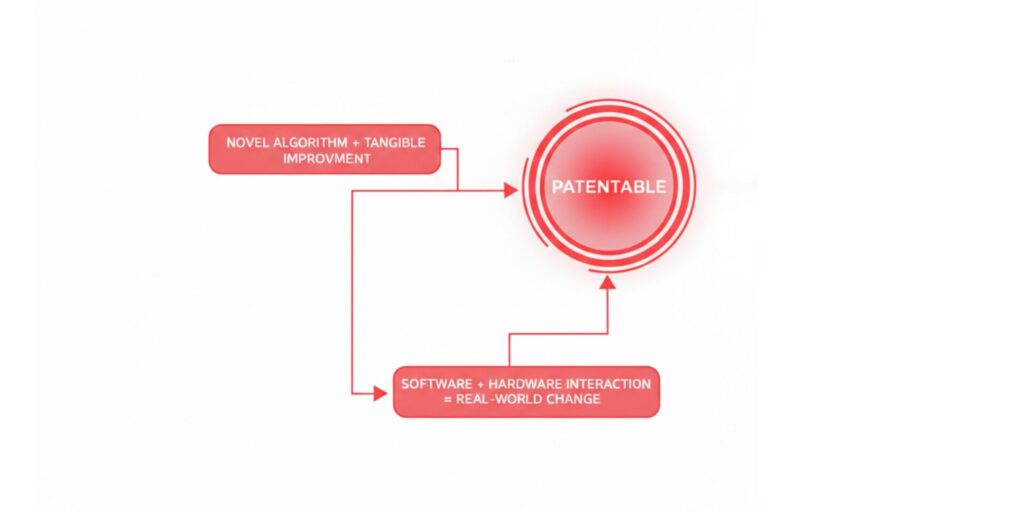


Determining Technical Effect in CRIs
Under the 2025 framework, software-related inventions are generally patentable if they result in:
- Faster processing speeds or reduced memory usage.
- Enhanced security protocols or improved data compression.
- The technical control of an external industrial or medical device (e.g., a robotic surgical arm).
- Improvements in signal transmission or network infrastructure.
A critical nuance in the 2025 guidelines is the treatment of AI. The IPO has specified that AI-generated inventions are not patentable because AI is not recognized as a “legal person” under the Patents Act, 1970. For an AI-related invention to be granted, the application must disclose the underlying architecture, training data characteristics, and parameters to meet the “sufficiency of disclosure” requirement. If the invention primarily mimics human thinking without a hardware-optimized technical benefit, it may be rejected as a “mental act” under Section 3(m).
Safeguarding Heritage: Section 3(p) and Traditional Knowledge
Section 3(p) was designed to prevent bio piracy the unauthorized patenting of traditional knowledge held by indigenous communities. The necessity for this provision was highlighted by high-profile international cases in the 1990s involving turmeric and neem.
In the “turmeric case,” two scientists at the University of Mississippi were granted a US patent for the use of turmeric powder in wound healing. India’s Council of Scientific and Industrial Research (CSIR) challenged the patent, providing evidence from ancient Sanskrit texts and scientific papers proving that turmeric’s medicinal properties were prior art. The US Patent and Trademark Office (USPTO) subsequently revoked the patent. Similarly, a patent on the fungicidal properties of neem was revoked by the European Patent Office (EPO) after India demonstrated its long-standing traditional use.
To provide a more systematic defence, India established the Traditional Knowledge Digital Library (TKDL) in 2001. The TKDL is a digital database that translates traditional Indian knowledge from ancient languages into a format accessible to global patent examiners. This allows examiners to identify traditional knowledge as “prior art” during the search phase, preventing the grant of wrongful patents.
Strategic Transformation of Section 4: Atomic Energy
Section 4 of the Act originally imposed an absolute bar on patents for any invention relating to atomic energy. This was based on national security concerns, ensuring that all nuclear technologies remained under strict State control. However, this blanket prohibition often stifled innovation in civilian and peaceful applications, such as nuclear medicine or safety sensors.
The landscape changed with the SHANTI Act (Sustainable Harnessing and Advancement of Nuclear Energy for Transforming Act, 2025). The amended Section 4 now allows the Central Government to grant patents for inventions relating to the “peaceful uses” of nuclear energy and radiation. This shift allows for private innovation in areas like radiation anomalies detection, vibration monitoring in facilities, and medical diagnostic tools using isotopes, while maintaining restrictions on strategic technologies like fuel enrichment.
The Indian Patent Filing Framework: A Step-by-Step Guide
Navigating the procedural requirements of the IPO is essential for innovators to secure and maintain their rights. The process is highly structured, with specific forms and strict timelines.
Types of Patent Applications
| Application Type | Description |
|---|---|
| Provisional Application | Filed when the invention is in the early stages of development. It secures an early "priority date" and provides a 12-month window to file the complete specification. |
| Complete Application | A detailed application containing the full specification, claims, and diagrams. |
| Ordinary Application | A non-provisional application filed for an invention that has not been filed anywhere else, primarily seeking protection in India. |
| Convention Application | Filed in India based on a prior filing in a Paris Convention member country. It must be filed within 12 months of the original filing to claim the same priority date. |
| PCT National Phase | An international application under the Patent Cooperation Treaty (PCT) that enters the Indian phase within 31 months of the earliest priority date. |
| Divisional Application | Filed if a single application contains multiple inventions. It can be filed voluntarily or at the direction of the Controller. |
| Patent of Addition | Filed for a slight modification or improvement of an existing patented invention. No separate renewal fee is required, and it expires with the main patent. |
The Procedural Lifecycle
The path from filing to grant involves several critical stages:
- Drafting and Filing: The application is filed using Form 1 (Application for Grant) and Form 2 (Specification). If the invention is not fully developed, a provisional specification is submitted.
- Publication: The application is typically published in the Official Journal 18 months after the filing or priority date. However, an applicant can request “early publication” using Form 9 to have the application published within one month.
- Request for Examination (RFE): Unlike many other jurisdictions, the IPO does not examine applications automatically. The applicant must file an RFE using Form 18 or Form 18A (for expedited examination). For applications filed on or after March 15, 2024, the deadline to file an RFE is 31 months from the priority date (reduced from 48 months).
- Examination and FER: An examiner reviews the application for novelty, inventive step, and industrial applicability. The examiner issues a First Examination Report (FER) detailing any objections.
- Response to FER: The applicant must respond to all objections in the FER within six months, with a possible three-month extension. If the objections are not addressed, the application is deemed abandoned.
- Grant: Once the Controller is satisfied that the invention meets all criteria, the patent is granted and recorded in the Register of Patents.
Summary
The Patents Act, 1970, has successfully navigated the complex journey from a process-only regime aimed at industrial survival to a TRIPS-compliant product patent system that ranks among the most sophisticated in the world. Its unique provisions, such as Section 3(d) and Section 3(p), represent a nuanced “middle path” that protects genuine innovation while safeguarding public health and traditional heritage. The recent 2024 and 2025 amendments reflect a forward-looking approach, emphasizing digitalization, procedural speed, and administrative adjudication over criminalization. As India continues to integrate into the global technology ecosystem, Intellect Bastion helps innovator to protect their invention under the Patents Act which remains the fundamental framework ensuring that the nation’s quest for innovation is both legally protected and socially responsible.




